For laboratory
Summary of Protoplast Preparation
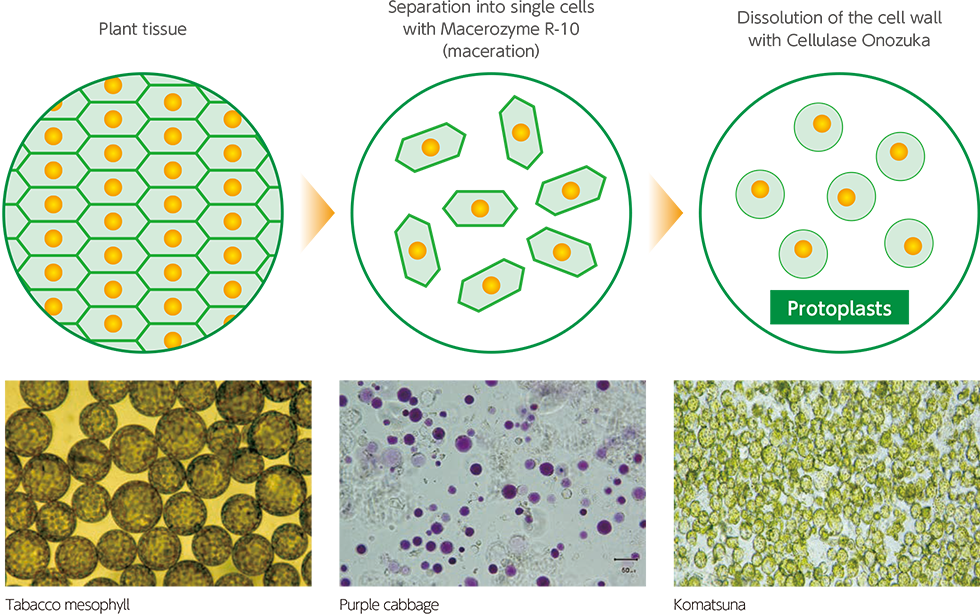
If plant tissue is separated into single cells with plant tissue macerating enzymes and then treated with plant tissue degrading enzymes, the plant tissue becomes a single cell, If it is furtherand treated with cell wall degrading enzymes, the cell wall dissolves leaving spherical cells encased in the cell membrane, known as protoplasts. These are used in cell engineering for a variety of different purposes, including; plotoplasts from different types of plants being fused together to create new plants, or new genes being introduced into cells to give plants new properties.
Illustration of Protoplast Preparation

Products
| 10g type(conventional product) | |||
|---|---|---|---|
| Product name | Cellulase Onozuka™ R-10 |
Cellulase Onozuka™ RS |
Macerozyme™ R-10 |
| Enzyme activity |
More than 10,000u/g |
More than 16,000u/g |
More than 3,000u/g |
| Packaging |  |
 |
 |
| 10g/brown bottle | |||
| Characteristic | Cellulase Onozuka R-10 and Cellulase Onozuka RS both degrade natural cellulose and dissolve plant cell walls. Used in isolating protoprasts from plant mesophyll and cultured cells. |
Macerozyme R-10contains pectinase and hemicellulase at high titers, and degrades plant tissues to single cells. | |
| Optimal pH | pH4.0-5.0 | pH5.0-6.0 | |
| Optimal temperature |
40-55℃ | 40-50℃ | |
small vials
Improved solubility
| Vial type | ||
|---|---|---|
| Cellulase Onozuka™ R-10 |
Cellulase Onozuka™ RS |
Macerozyme™ R-10 |
| More than 10,000u/g |
More than 16,000u/g |
More than 3,000u/g |
 |
 |
 |
| 5 vials/box | ||
| Cellulase Onozuka R-10 and Cellulase Onozuka RS both degrade natural cellulose and dissolve plant cell walls. Used in isolating protoprasts from plant mesophyll and cultured cells. |
Macerozyme R-10contains pectinase and hemicellulase at high titers, and degrades plant tissues to single cells. | |
| pH4.0-5.0 | pH5.0-6.0 | |
| 40-55℃ | 40-50℃ | |
Vial type is Prepackaged in Small Vials
Packaging in small vials filled with a volume equivalent to 1 g of the conventional product eliminates the need for weighing when preparing a solution.

Excellent Solubility of Vial type
Improved solubility compared to the conventional product, enhances workability and operability.


History of Protoplast
Why are protoplasts produced?
The characteristic feature of plant cells is said to be the presence of cell walls, which constitute building blocks for organizing plant body. Thus the cell walls are important for plant structure. However, as the cell walls are outcomes of the vital activities of protoplasts, the protoplasts are ultimately more important than cell walls.
Plant protoplasts were first recognized in the first half of the 19th century, when plant cells were placed in the hypertonic medium and the resultant cellular entity of the protoplasts within the cell walls was observed. These observations were closely linked to the development of cell theory, which was established around the same time.
These early observations led naturally to attempt to isolate protoplasts and cultivate them under artificial conditions. In 1909, Ernst Küster at Gießen, Germany, isolated protoplasts squeezed from the cut end of cell walls with aid of a sharp razor; however, the resultant amount of protoplasts was far below what was needed for experimental works1).In 1960, Edward C. Cocking at Nottingham, UK, isolated protoplasts from tomato roots using a cellulase extracted and purified from the culture filtrates of a wood rotting fungus of Myrothesium verrucaria 2). This approach of using a cell wall lytic enzyme to isolate plant protoplasts was methodologically advanced; however, the quantity of protoplasts that could be obtained was still low. It required researchers to culture the fungus and isolate the enzyme as part of experimental protocols.
Tobacco mesophyll protoplasts
In 1968, Takebe et al. successfully isolated protoplasts from tobacco leaves with aid of Cellulase Onozuka R10 and Macerozyme R10, produced by Yakult Pharmaceutical Co., Ltd. (at that time Kinki Yakult Manufacturing Co., Ltd.) 3). Macerozyme is prepared from Rhizopus sp., while Cellulase Onozuka is prepared from the deuteromycete Trichoderma viride. Preparation of the isolated protoplasts consisted of two steps: first the degradation of pectic substances in leaf tissues by Macerozyme to isolate the cells, from which the protoplasts are isolated by use of Cellulase Onozuka. A single-step preparation method is also available, in which these two enzymes are mixed.
As a result, tens of millions of protoplasts are obtained from a single leaf (Figure 1). Subsequently, Takebe and Otsuki established a one-step growth system for culture system for infection of these protoplasts with tobacco mosaic virus (TMV) 4), resulting in the elucidation of the molecular mechanisms of the early stages of TMV infection. The one-step growth system was originally developed for synchronous viral propagation by infecting bacteria with phages,which contributes brilliant outcomes in the early stage of molecular biology, as is vividly described in a book on profile about Max Delbrück 5). Although TMV was crystallized as the first case among viruses, molecular study of TMV propagation was undeveloped by the time because of the lack of such a one-step growth system.
This study became the trigger for the further development of plant protoplast research.
Figure 1 Tobacco mesophyll protoplasts

A leaf has about 5 to 7 × 107 mesophyll cells from which protoplasts can be prepared. Protoplasts can also be obtained from epidermal cells and guard cells, depending on the techniques used.
Plant regeneration from protoplasts
By the late 1960s many attempts had been made to culture isolated plant protoplasts, but without success.
The idea of culturing leaf cells can be traced to a proposal by Gottlieb Harberlandt at Linz, Austria, in 1902 6), but even in 1964, the monograph on cell and tissue culture authored by Philip R. White, a notable pioneer in this field, stated that it would be almost impossible to culture leaf cells 7). Nonetheless, Nagata and Takebe were ultimately able to isolate from the mesophyll of tobacco leaves and demonstrate unambiguously that not only were mesophyll protoplasts suitable for culture, but the process of cell division was synchronous when properly prepared starting materials were selected for culturing isolated tobacco mesophyll protoplasts 8). Cultured cell populations derived from tobacco mesophyll cells initiated DNA synthesis at 12 hours, started cell division at 24 hours (Figure 2), and cell division was completed in almost all cells by 72 hours. This created the possibility of culturing mesophyll protoplasts for cell biological studies of all kinds. Nagata and Takebe also demonstrated that proper selection of culture conditions and the use of agar media lead to colony formation at high frequency, indicating that higher plant cells can be handled almost like microorganisms (Figure 3).
After the colonies obtained from mesophyll protoplasts were transferred to conditions appropriate for organ differentiation, it was also shown for the first time that the regeneration of whole plants from any of these colonies is feasible (Figures 4 and 5). Although the totipotency of plant cells had been shown in the early 1960s, this culture system for tobacco mesophyll protoplasts was the first demonstration that the totipotency of somatic cells id almost 100% 9). This method was soon applied to plant regeneration from protoplasts obtained from potato leaves, and the obtained plants were reported to retain enough genetic diversity to be useful for the purposes of potato breeding 10). Since then, protoplasts have been prepared from a variety of plant species, and tissue studies and plant regeneration experiments have become a routine and extensively performed approach in the plant science research.
Around the same time as these key experiments, it was discovered that cell fusion can d be induced between plant protoplasts treated with polyethylene glycol, thereby leading to the beginning of a new area of“ somatic cell hybrid genetics.” The production of pomato and topato, which are somatic hybrids of potato and tomato 11), marked the beginning of plant biotechnology and a wide range of further developments 12). An overview of the growth of plant biotechnology research stemming from these advances can be found in many textbooks 13).
Figure 2 Cell division induced by protoplasts
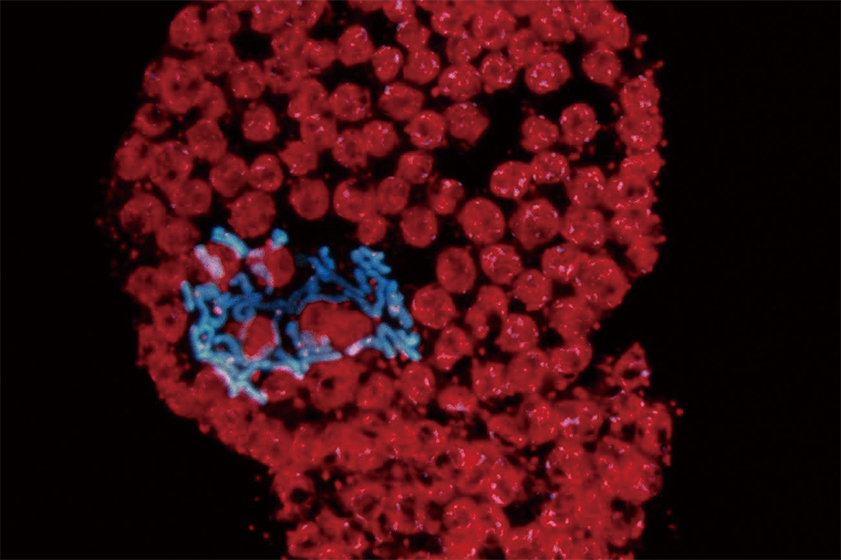
Chromosomes are clearly visible and appear in blue using DAPI staining and ultraviolet illumination. Chloroplasts are visible in red.
Figure 3 Tobacco mesophyll protoplasts forming colonies on agar media
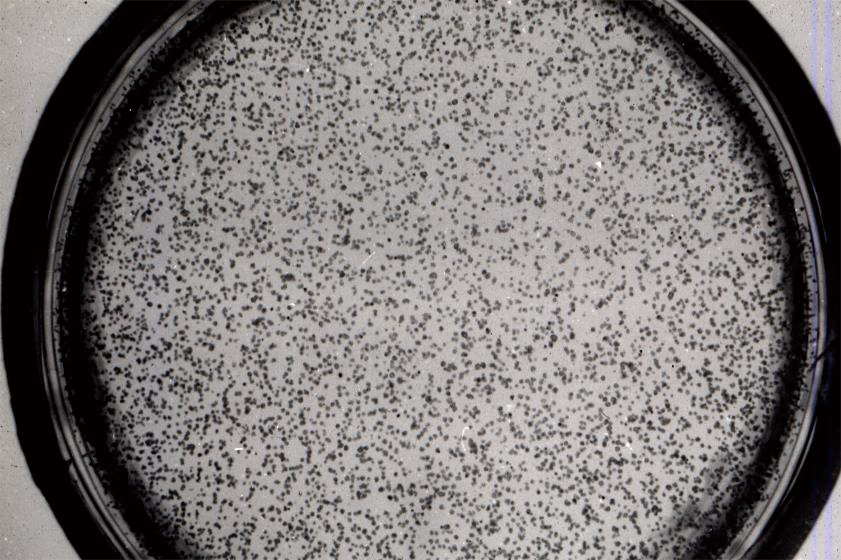
A total of 60% to 80% of protoplasts plated on agar media form colonies.
Colony formation is dependent on cell density, and a culture of at least 1000 cells/mL is required.
Figure 4 Development of organs from colonies
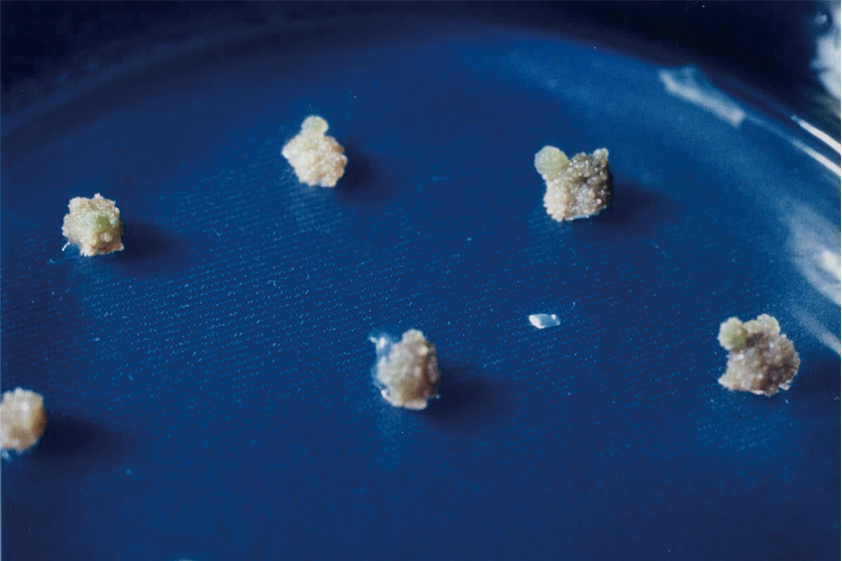
Each colony differentiates to form shoots followed by roots and then seedlings when the concentration ratios of auxin and cytokinin are set at the proper levels for organ differentiation.
Figure 5 The first plants that were regenerated from a single mesophyll protoplast

This was the plant regenerated as described above and photographed in April 1971.
Further challenges in protoplast preparation
Although the use of Macerozyme R10 and Cellulase Onozuka R10 brought a new dimension to plant sciences, enzymatic treatment is an artificial procedure that works best if it is kept as simple and brief as possible. In that sense, the use of additional new enzymes was explored, such as Pectolyase Y-23 (produced initially by Seishin Pharmaceutical Co., Ltd., and currently by Kyowa Kasei Co., Ltd.), which is used for soy sauce production. This enzyme, which is obtained from the mold Aspergillus japonicus, is composed of polygalacturonase and pectin lyase along with activators of both enzymes, and it permitted cell isolation from a broader range of plants 14). Around that time, the tobacco BY-2 cell line was being widely studied as a model plant cell line, and it became used worldwide as a system with a highly synchronous cell population, coming to be known as the HeLa cell line of plant cell biological research 15). Cellulase Onozula R10, however, proved insufficient for protoplast preparation from this cell line and it was found that protoplast formation from BY-2 cells required the use of a newly developed enzyme, Cellulase Onozuka RS (Yakult Pharmaceutical Industry Co., Ltd.) 16). A combination of Pectolyase Y-23 and Cellulase Onozuka RS is now proven to widely applicable, which has opened the door for the preparation of protoplasts from much wider range of plant tissues and plant organs, in particular from rice leaf sheaths which had been resistant to other enzymes 14).
Protoplasts as materials for transformation
Transformation is an essential technique in modern plant sciences. Because protoplast transformation is possible by means of DNA introduction, transformation became widely used as a means for gene transfer. Therefore, protoplasts were prepared from a variety of cells for this purpose.
In particular, since it was initially thought that rice plants cannot be transformed by Agrobacterium tumefaciens, rice protoplasts were used as materials for transformation.
In fact, plant regeneration using protoplasts from rice embryos was successfully achieved and was used for transformation 17). Because it was later shown that rice cells can be transformed by Agrobacterium tumefaciens 18), the use of rice protoplasts became less important, but they have continued to be used for certain purposes. In addition, protoplasts have been widely used for studying transient gene expression, and they are now an essential means for the confirmation of expression from geneexpression constructs, a routine procedure in modern plant sciences.
Relevance of experimental protoplast systems
In contrast to the early days when protoplast preparation methods were being developed using a variety of plant materials and a wide varieties of results had been published in scientific journals, the situation today is less dynamic. However, there are still experimental systems in which the use of isolated protoplasts is inevitable.
Plant cells are cytologically characterized by the presence of vacuoles that are assumed to have various functions, but because cells without vacuoles did not naturally exist, these functions remained speculative and difficult to investigate. However, it was found that when tobacco BY-2 protoplasts were placed under ultracentrifugal field, vacuoles with low specific gravity could be expelled from the protoplasts (which contain cytoplasm and nuclear fractions with high specific gravity). In this way, cells without vacuoles could be made available for experimentation 19).
These miniprotoplasts without vacuoles exhibit a variety of interesting behaviors, and in vitro propagation of tomato mosaic virus (ToMV) was demonstrated when ToMV—closely related to TMV—was added to cell-free extracts prepared from such miniprotoplasts 20). Although in vitro replication systems for animal viruses in animal cells have a long history, this was the first in vitro growth system established for viral replication in plants. This provides clear evidence that the presence of vacuoles in plant cells prevents the preparation of such in vitro systems for virus propagation. The availability of these experimental systems may lead to the elucidation of the detailed molecular mechanisms underlying the propagation of plant viruses.
An interesting development is that the propagation of cucumber mosaic virus (CMV), but not TMV, was observed in protoplasts from cultured rice cells transfected with TMV-RNA or CMV-RNA, possibly due to the inability of TMV propagation in rice cells. However, in rice protoplasts co-transfected with TMV-RNA and CMV-RNA, TMV propagation was observed in cells with CMV propagation.
This suggested that CMV propagation suppressed the inhibition of TMV propagation that is usually observed in rice cells, and hence permits TMV propagation 21). Although the mechanism remains to be elucidated, this was the first system in plant cells in which the inhibition of viral propagation and then the subsequent removal of that inhibition, are observed.
This system may prove highly valuable for elucidating the molecular mechanisms of microRNA, a field in which rapid advances are being made.
Conclusion
Protoplast research has been widely developed as a key methodology for plant biotechnology, and certain experimental systems that are important in modern plant sciences t may not have been feasible without the ability to work with isolated protoplasts. Information obtained from the use of such systems have made fundamental contributions to plant sciences. It has already been 50 years and 36 years since the introduction of Macerozyme R10 and Cellulase Onozuka R10, and Cellulase Onozuka RS, respectively, all of which are produced by Yakult Pharmaceutical Co., Ltd., for use in the preparation of plant protoplasts. Such long-lived research methods are rare in the rapidly progressing field of life sciences. As a person involved in this field from its early days, this author was moved to write this paper to share these facts and place them in the broader content of the history of the plant science.Contributed in July 2018
References
-
1) Küster, E.:Ber. Deutsch. Bot. Gesell. 27,589(1909)
-
2) Cocking, E.C. :Nature 187, 962(1960)
-
3) Takebe, I., Otsuki, Y., Aoki, S. :Plant Cell Physiol. 9, 115(1968)
-
4) Takebe, I., Otsuki, Y.:Proc. Natl. Acad. Sci. USA 84, 643(1969)
-
5) Fischer, E.P., Lipson, C.: Thinking about Science - Max Delbrück and the Origin of Molecular Biology, W.W. Norton & Co.(1988)
-
6) Harberlandt, G.:In:Plant Tissue Culture - 100 Years since Gottlieb Harberlandt, Laimer, M,, Rücker, W. (eds.), Springer-Verlag, Wien(2002)
-
7) White, P.R.: The Cultivation of Animal and Plant Cells, Ronald Press(1964)
-
8) Nagata, T., Takebe, I. :Planta 92, 201(1970)
-
9) Nagata, T., Takebe, I:Planta 99, 12(1971)
-
10) Melchers, G., Sacristán, M.D., Holder, A.A.:Carlsberg Res. Comm. 44, 127(1979)
-
11) Nagata, T., Bajaj, Y.P.S.(eds.): Biotechnology in Agriculture and Forestry, Vol. 49, Springer-Verlag, Heidelberg(2002)
-
12) Shephard, J., Bidney, D., Shahin, E. :Science 208, 17(1980)
-
13) Nagata, T., Cell Engineering of Plant Protoplasts, Kodansha Sci entific, Tokyo (1993) (in Japanese)
-
14) Nagata, T., Ishii, S. :Can. J. Bot. 57, 1820(1979)
-
15) Nagata, T., Nemoto, Y., Hasezawa, S.: Int. Rev. Cytol. 132, 1(1992)
-
16) Nagata, T., Okada, K., Takebe, I., Matsui, C. :Mol. Gen. Genet. 184, 161(1981)
-
17) Shimamoto, K., Terada, R., Izawa, T., Fujimoto, H. :Nature 338, 274(1989)
-
18) Hiei, Y., Komari, T., Kubo, T.: Plant Mol. Biol. 35, 205(1997)
-
19) Sonobe, S. :Protoplasma 155, 239(1990)
-
20) Komoda, K., Naito, S., Ishikawa, M.:Proc. Natl. Acad. Sci. USA 101, 1863(2004)
-
21) Okada, K., Nagata, T., Takebe, I.:Plant Cell Rep.7, 333(1988)
